Use these charts to guide proper dosage for your patients 12 and older
Jump to product:
- TYLENOL® PRECISE™ Pain Relieving Cream
- TYLENOL® PRECISE™ Cooling Pain Relieving Cream
- TYLENOL® Regular Strength Liquid Gels
- TYLENOL® Regular Strength Tablets
- TYLENOL® Family, Children & Adults Oral Suspension
- TYLENOL® Extra Strength Caplets
- TYLENOL® Extra Strength Coated Tablets
- TYLENOL® Extra Strength Dissolve Packs
- TYLENOL® Rapid Release Gels
- TYLENOL® 8HR Arthritis Pain Tablets
- TYLENOL® 8HR Muscle Aches & Pain Tablets
- TYLENOL® PM Extra Strength Caplets
Patients may have questions about over-the-counter analgesics. Use the information below to help them find the TYLENOL® product that’s right for them. Remember, many medicines contain the same active ingredient, so remind patients to use ONLY 1 medicine containing the same active ingredient at a time.
PROFESSIONAL DISCRETIONARY DOSING: To help encourage the safe use of acetaminophen, in 2011, the makers of TYLENOL® lowered the labeled maximum daily dose for single-ingredient TYLENOL® Extra Strength (acetaminophen) products sold in the US from 8 pills/day (4000 mg) to 6 pills/day (3000 mg). The dosage interval also changed from 2 pills every 4 to 6 hours to 2 pills every 6 hours. If pain or fever persists at the total labeled daily dose, healthcare professionals may exercise their discretion and recommend up to 4000 mg/day.*
*The efficacy and safety of TYLENOL® at 4000 mg/day are well established.
| TYLENOL® PRECISE™ Pain Relieving Cream | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE & DIRECTIONS | DOSAGE FREQUENCY | DIRECTIONS |
|
Lidocaine 4% |
Not more than 3 to 4 times daily |
Adults and children 12 years and over:
Children under 12 years:
FOR EXTERNAL USE |
|
|
Stop use and ask a doctor if condition worsens or symptoms persists for more than 7 days, and if symptoms clear up but occur again within a few days. |
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. |
||
| TYLENOL® PRECISE™ Cooling Pain Relieving Cream | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE & DIRECTIONS | DOSAGE FREQUENCY | DIRECTIONS |
|
Lidocaine 4% Menthol 1% |
Not more than 3 to 4 times daily |
Adults and children 12 years and over:
Children under 12 years:
FOR EXTERNAL USE |
|
|
Stop use and ask a doctor if condition worsens or symptoms persists for more than 7 days, and if symptoms clear up but occur again within a few days. |
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. |
||
| Regular Strength TYLENOL® Regular Strength Liquid Gels | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE* & DIRECTIONS | DOSAGE FREQUENCY* | DIRECTIONS |
|
Acetaminophen 325 mg (in each capsule) |
2 capsules every 4-6 hours while symptoms last |
Not to exceed 10 capsules in 24 hours, unless directed by a doctor Total labeled daily dose: |
|
|
PROFESSIONAL DISCRETIONARY DOSAGE: If pain or fever persists at the total labeled daily dose, healthcare professionals may exercise their discretion and recommend up to 4000 mg/day.† |
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
||
*Consult TYLENOL® Regular Strength liquid gels packaging for dosing children under 12 years.
†The efficacy and safety of TYLENOL® at 4000 mg/day are well established.
| Regular Strength TYLENOL® Regular Strength Tablets | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE* & DIRECTIONS | DOSAGE FREQUENCY* | DIRECTIONS |
|
Acetaminophen 325 mg (in each tablet) |
2 tablets every 4-6 hours while symptoms last |
Not to exceed 10 tablets in 24 hours, unless directed by a doctor Total labeled daily dose: |
|
|
PROFESSIONAL DISCRETIONARY DOSAGE: If pain or fever persists at the total labeled daily dose, healthcare professionals may exercise their discretion and recommend up to 4000 mg/day.† |
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
||
*Consult TYLENOL® Regular Strength tablet packaging for dosing children under 12 years.
†The efficacy and safety of TYLENOL® at 4000 mg/day are well established.
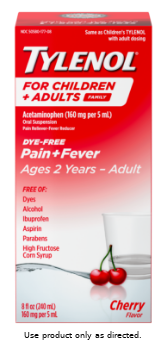
| TYLENOL® Family, Children & Adults Oral Suspension | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT | DOSAGE FREQUENCY | DIRECTIONS |
|
Acetaminophen 160 mg (in each 5 mL) |
Take every 4 hours while symptoms last | Not to exceed 5 doses in 24 hours, unless directed by a doctor | |
| IF POSSIBLE, USE WEIGHT TO DOSE; OTHERWISE, USE AGE. | |||
| WEIGHT | AGE | DOSE* | |
| Under 24 lbs | Under 2 years | Ask a doctor | |
| 24-35 lbs | 2-3 years |
5 mL
 |
|
| 36-47 lbs | 4-5 years |
7.5 mL
 |
|
| 48-59 lbs | 6-8 years |
10 mL
 |
|
| 60-71 lbs | 9-10 years |
12.5 mL
 |
|
| 72-95 lbs | 11 years |
15 mL
 |
|
| 96 lbs and over | Adults and children 12 years and over |
20 mL
  |
|
|
PROFESSIONAL DISCRETIONARY DOSAGE: If pain or fever persists at the total labeled daily dose, healthcare professionals may exercise their discretion and recommend up to 4000 mg/day.† Note: For children under 12 years, the maximum daily amount is 5 doses in 24 hours. |
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
||
mL = milliliter
*Or as directed by a doctor.
†The efficacy and safety of TYLENOL® at 4000 mg/day are well established.
| Extra Strength TYLENOL® Extra Strength Caplets | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE* & DIRECTIONS | DOSAGE FREQUENCY* | DIRECTIONS |
|
Acetaminophen 500 mg (in each caplet) |
2 caplets every 6 hours while symptoms last |
Not to exceed 6 caplets in 24 hours, unless directed by a doctor Total labeled daily dose: |
|
|
PROFESSIONAL DISCRETIONARY DOSAGE: If pain or fever persists at the total labeled daily dose, healthcare professionals may exercise their discretion and recommend up to 4000 mg/day.† |
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
||
*For children under 12 years, at healthcare professional's discretion.
†The efficacy and safety of TYLENOL® at 4000 mg/day are well established.
| Extra Strength TYLENOL® Extra Strength Coated Tablets | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE* & DIRECTIONS | DOSAGE FREQUENCY* | DIRECTIONS |
|
Acetaminophen 500 mg (in each tablet) |
2 tablets every 6 hours while symptoms last |
Not to exceed 6 tablets in 24 hours, unless directed by a doctor Total labeled daily dose: |
|
|
PROFESSIONAL DISCRETIONARY DOSAGE: If pain or fever persists at the total labeled daily dose, healthcare professionals may exercise their discretion and recommend up to 4000 mg/day.† |
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
||
*For children under 12 years, at healthcare professional's discretion.
†The efficacy and safety of TYLENOL® at 4000 mg/day are well established.
| Extra Strength TYLENOL® Extra Strength Dissolve Packs | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE* & DIRECTIONS | DOSAGE FREQUENCY* | DIRECTIONS |
|
Acetaminophen 500 mg (in each powder) |
2 powders every 6 hours while symptoms last |
Not to exceed 6 powders in 24 hours, unless directed by a doctor Total labeled daily dose: |
|
|
PROFESSIONAL DISCRETIONARY DOSAGE: If pain or fever persists at the total labeled daily dose, healthcare professionals may exercise their discretion and recommend up to 4000 mg/day.† |
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
||
*For children under 12 years, at healthcare professional's discretion.
†The efficacy and safety of TYLENOL® at 4000 mg/day are well established.
| Extra Strength TYLENOL® Rapid Release Gels | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE* & DIRECTIONS | DOSAGE FREQUENCY* | DIRECTIONS |
|
Acetaminophen 500 mg (in each gelcap) |
2 gelcaps every 6 hours while symptoms last |
Not to exceed 6 gelcaps in 24 hours, unless directed by a doctor Total labeled daily dose: |
|
|
PROFESSIONAL DISCRETIONARY DOSAGE: If pain or fever persists at the total labeled daily dose, healthcare professionals may exercise their discretion and recommend up to 4000 mg/day.† |
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
||
*For children under 12 years, at healthcare professional's discretion.
†The efficacy and safety of TYLENOL® at 4000 mg/day are well established.
| Extended Release TYLENOL® 8HR Arthritis Pain Tablets | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE* & DIRECTIONS | DOSAGE FREQUENCY* | DIRECTIONS |
|
Acetaminophen 650 mg (in each bi-layer tablet) |
2 bi-layer tablets every 8 hours with water |
Not to exceed 6 bi-layer tablets in 24 hours Total labeled daily dose: |
|
|
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
|||
*For children under 18 years of age, at healthcare professional’s discretion.
| Extended Release TYLENOL® 8HR Muscle Aches & Pain Tablets | |||
|---|---|---|---|
 |
ACTIVE INGREDIENT ACTIVE INGREDIENT, DOSAGE* & DIRECTIONS | DOSAGE FREQUENCY* | DIRECTIONS |
|
Acetaminophen 650 mg (in each bi-layer tablet) |
2 bi-layer tablets every 8 hours with water |
Not to exceed 6 bi-layer tablets in 24 hours Total labeled daily dose: |
|
|
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
|||
*Do not use in children under 12 years of age.
| PM Extra Strength TYLENOL® PM Extra Strength Caplets | |||
|---|---|---|---|
 |
ACTIVE INGREDIENTS ACTIVE INGREDIENTS, DOSAGE* & DIRECTIONS | DOSAGE FREQUENCY* | DIRECTIONS |
|
Acetaminophen 500 mg (in each caplet) |
2 caplets at bedtime |
Not to exceed 2 caplets in 24 hours Total labeled daily dose: |
|
|
IMPORTANT INSTRUCTIONS FOR PROPER USE: Read and follow the label on all TYLENOL® products. Do NOT use with any other product containing acetaminophen. |
|||
*Do not use in children under 12 years of age.
This is not a complete list.

Register
All Fields required, unless otherwise indicated
Personal Information
Step 1
Account Information
Step 2
Will be used as your user name
Address Information
Step 3
By submitting your information above, you agree that the information you provide will be governed by our site's Privacy Policy.